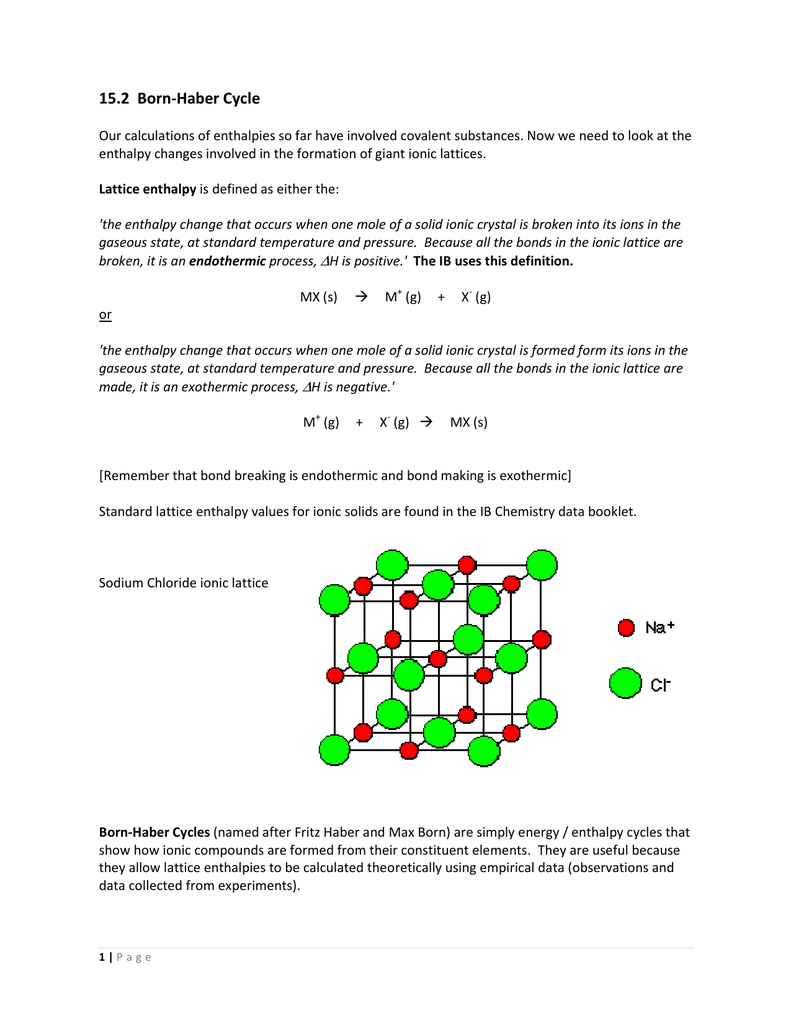
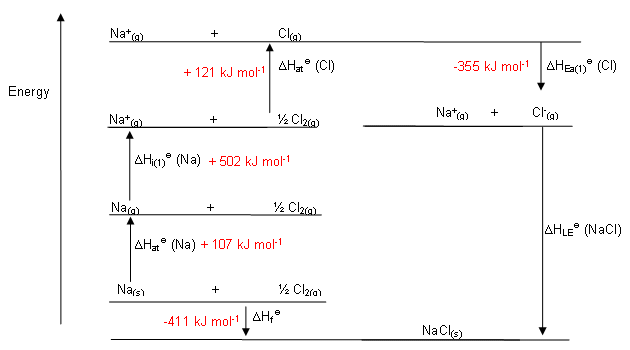
It follows, therefore, that the greater the lattice energy, numerically, the greater the energy required to break up the crystal into its constituent ions. In this way it is seen that the lattice energy is the same in magnitude as, but opposite in sign to, the energy of dissociation of the crystal.
Born haber cycle problems pdf free#
That is, from separated ions free from any solvent. The lattice energy is the change in energy that occurs when an ionic solid is separated into isolated ions in the gas phase.Īnother definition of the lattice energy is to regard it as the energy of formation of a given quantity of the crystal, e.g., 1 mole, from gaseous ions. The decrease in energy accompanying the process of bringing the ions, when separated from each other by an infinite distance, to the positions they occupy in the stable lattice. The energy released when a crystalline solid forms from ions is related to the lattice energy of the solid. The positive and negative ion in an ionic crystal are held together by electrostatic forces. Introduction of Lattice Energy Lattice energy, also called lattice enthalpy, of an ionic solid/crystal is a measure of the strength of bonds in that ionic compound. Inorganic Chemistry (2nd Edition) by Catherine E. Essentials of Physical Chemistry by Arun Bahl, B.S. General Chemistry (9th Edition) by Darrell D. Solid State Chemistry and its Applications by Anthony R West 03. Textbook of Physical Chemistry by Samuel Glasstone 02. Table 4 Representative Calculated Lattice Energies 13 Table 5 Experimental & Theoretical Lattice Energies in Kcal perĠ1. Tables Table 1 Selected Enthalpies of Sublimation at 298 K 9 Table 2 Selected Bond Dissociation Enthalpies at 298 K 10 Table 3 Summary of Reactions in the BornHaber Cycle for the 01 Introduction of Lattice Energy 02 02 Factors Affecting Lattice Energy of a Crystal 03 03 Attraction & Repulsion in a Crystal 04 04 The Relationship between Lattice Energies and PhysicalĠ5 Determination of Lattice Energy 06 06 The Born-Haber Cycle 07 07 Born-Haber Cycle: Explanation with an Examplesīorn-Haber Cycle for Sodium Chloride Born-Haber Cycle for Cesium Fluoride Born-Haber Cycle for Lithium FluorideĠ8 Importance of Born-Haber Cycle 13 09 Applications of Lattice Energy Determination 13 10 Problem 14
